WGCC Best Practices for Spiking RTU Bags
Spikes on IV administration sets can differ in size/dimensions between different manufacturers. Those with narrow, smaller spikes (for example; BD AlarisTM and Baxter CLEARLINK TM), should be completely inserted, depending on spike set used.Read More
New 2-gram Meropenem for Injection, USP Vial Exclusive from WG Critical Care, LLC
December 5, 2023Essential Injectable, Essential Injectable Product Updates, News & Announcements, Products
Meropenem is the sixth largest generic anti-infective product in unit volume in hospital and alternate site settings. According to a 2022 IQVIA report, the market value for Meropenem is $88.7 million dollars, with unit volume at 15.9 million annually.Read More
STATEMENT ON USE OF MEDICATIONS FOR CAPITAL PUNISHMENT
WG Critical Care, LLC is a healthcare company that is dedicated to improving the outcome of patients treated by doctors and other healthcare professionals in critical care facilities throughout the United States. As such, WG Critical Care does not condone the off-label use or misuse of its drugs including the use of its products to...Read More
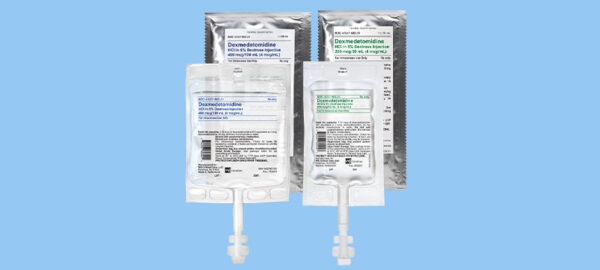
DHP Letter – Dexmedetomidine HCl in 5% Dextrose premix bag 200mcg/50mL (4mcg/mL) – Change in bag size
Dear Healthcare Professional,This letter is to inform you about an upcoming change on the bag size of WG Critical Care’s 200mcg/50mL (4mcg/mL) Dexmedetomidine HCl in 5% Dextrose premix bag. The fill volume will not change; however, the bag size will be smaller.Read More
WG Critical Care Announces First and Only FDA-Approved, Ready-To-Use Midazolam in 0.9% Sodium Chloride Injection CIV
PARAMUS, UNITED STATES, August 4, 2021 /EINPresswire.com/ — WG Critical Care, LLC will be introducing the first and only FDA-approved, Ready-To-Use (RTU) Midazolam in 0.9% Sodium Chloride Injection CIV in single-dose bags. Midazolam is currently controlled in schedule IV of the Controlled Substance Act. The launch of Ready-To-Use Midazolam in 0.9% Sodium Chloride Injection CIV...Read More